The real cost of clinical trials.
It’s no mystery that it costs pharmaceutical companies a very pretty penny to conduct research with the hope of getting investigational drugs approved. From drug development to site selection to protocol adherence, a sponsor invests millions and even billions of dollars to bring a drug to market. And this investment hinges on keeping patients enrolled for the full duration of the trial. To better understand how important it is to keep each and every patient engaged from start to finish, explore our infographic.

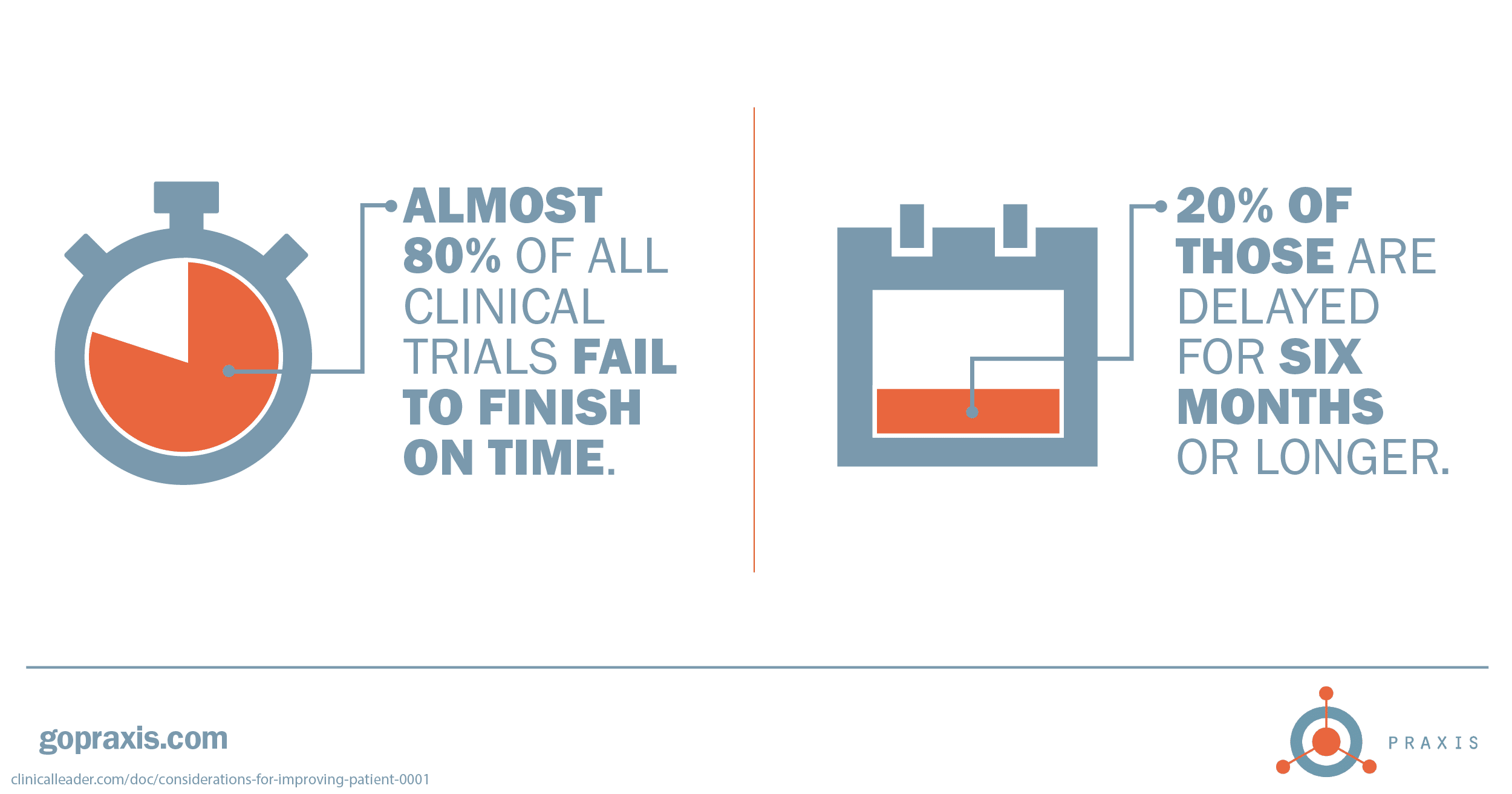

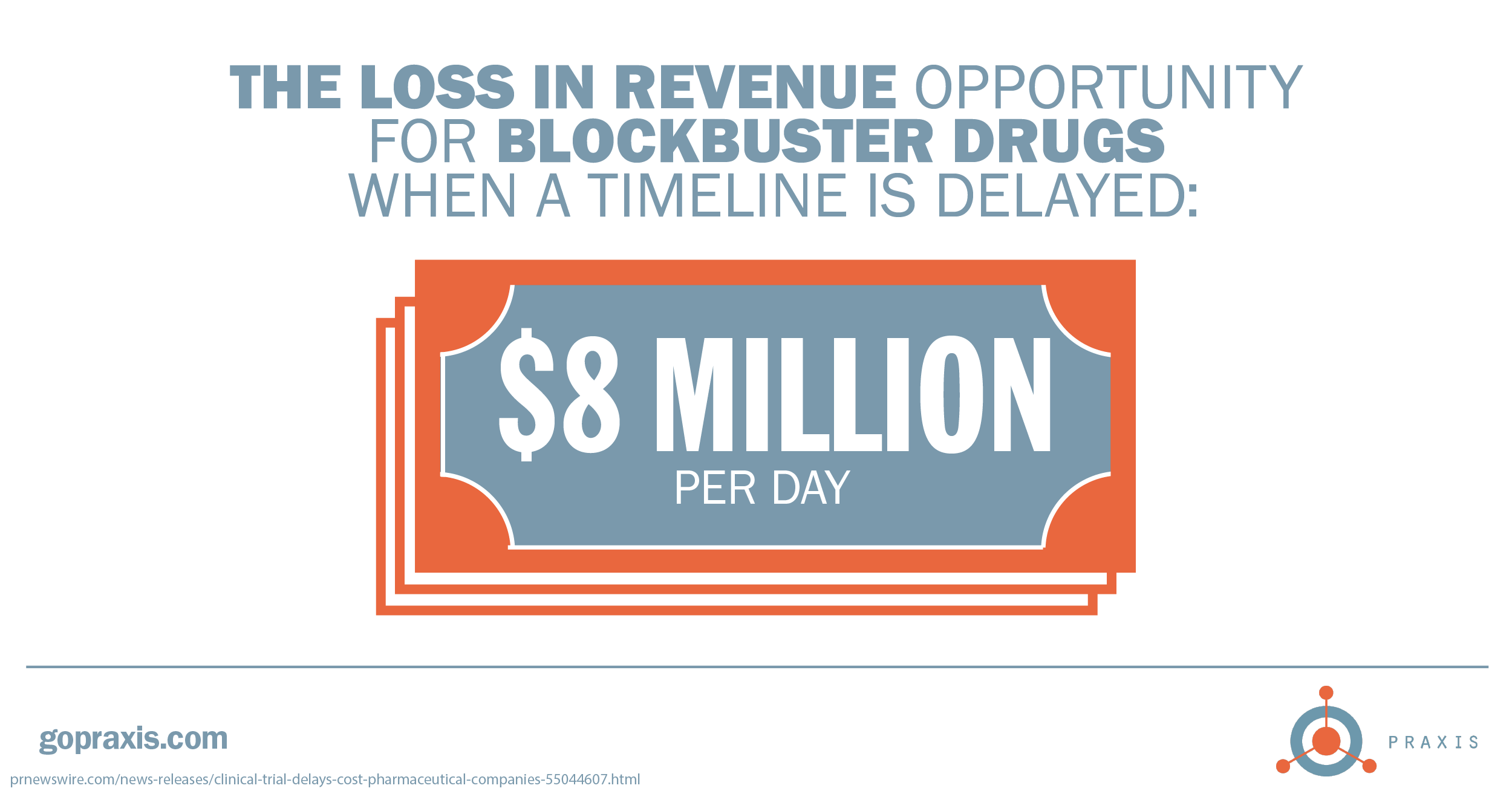
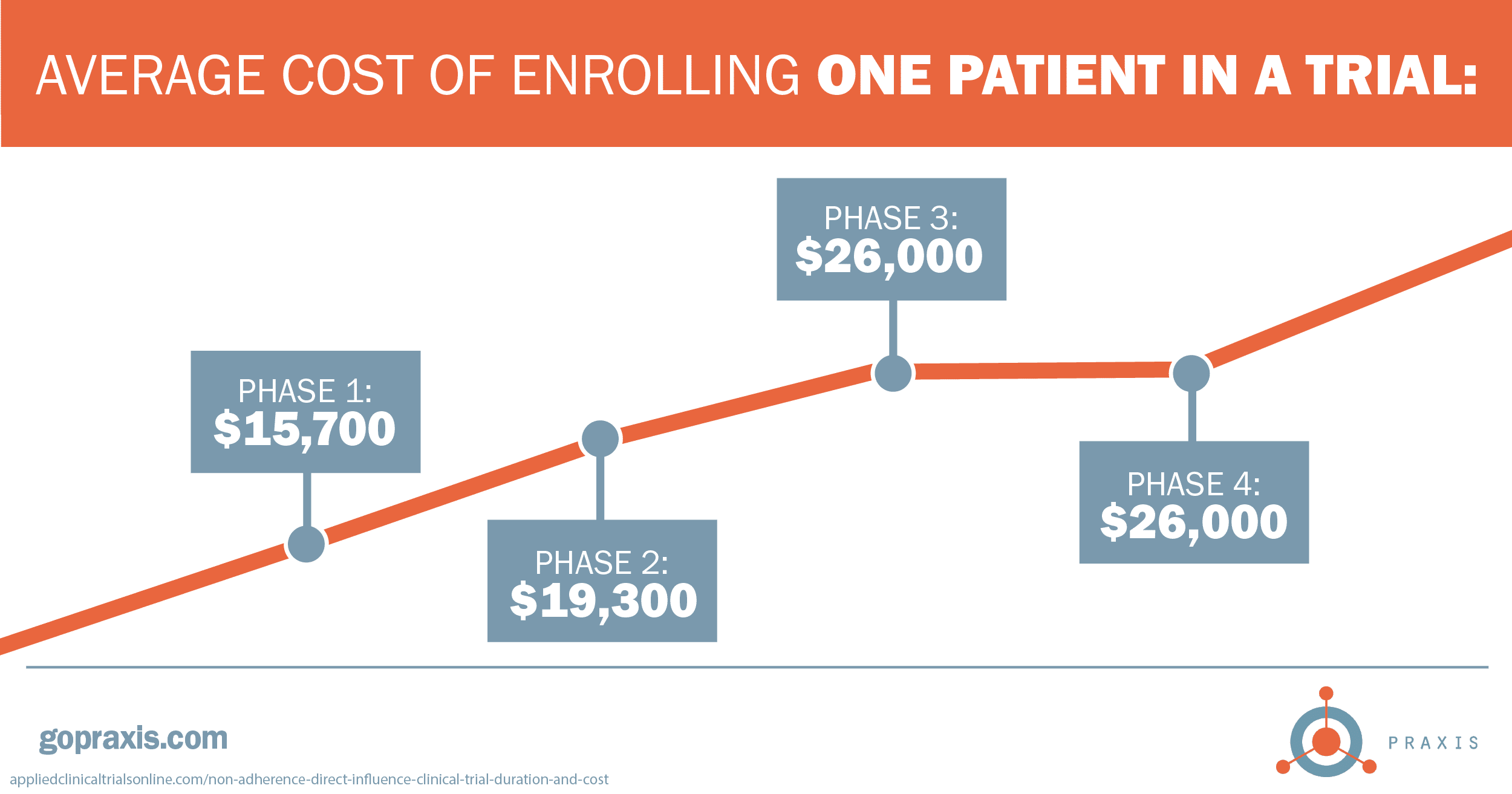

By the numbers, patient adherence and retention should be a high priority. Looking for some ideas to keep your patients engaged through trial completion? Drop us a line.
1 appliedclinicaltrialsonline.com/non-adherence-direct-influence-clinical-trial-duration-and-cost
2 clinicalleader.com/doc/considerations-for-improving-patient-0001
3 prnewswire.com/news-releases/clinical-trial-delays-cost-pharmaceutical-companies-55044607.html
4 prnewswire.com/news-releases/clinical-trial-delays-cost-pharmaceutical-companies-55044607.html
5 appliedclinicaltrialsonline.com/non-adherence-direct-influence-clinical-trial-duration-and-cost
6 clinicalleader.com/doc/considerations-for-improving-patient-0001

